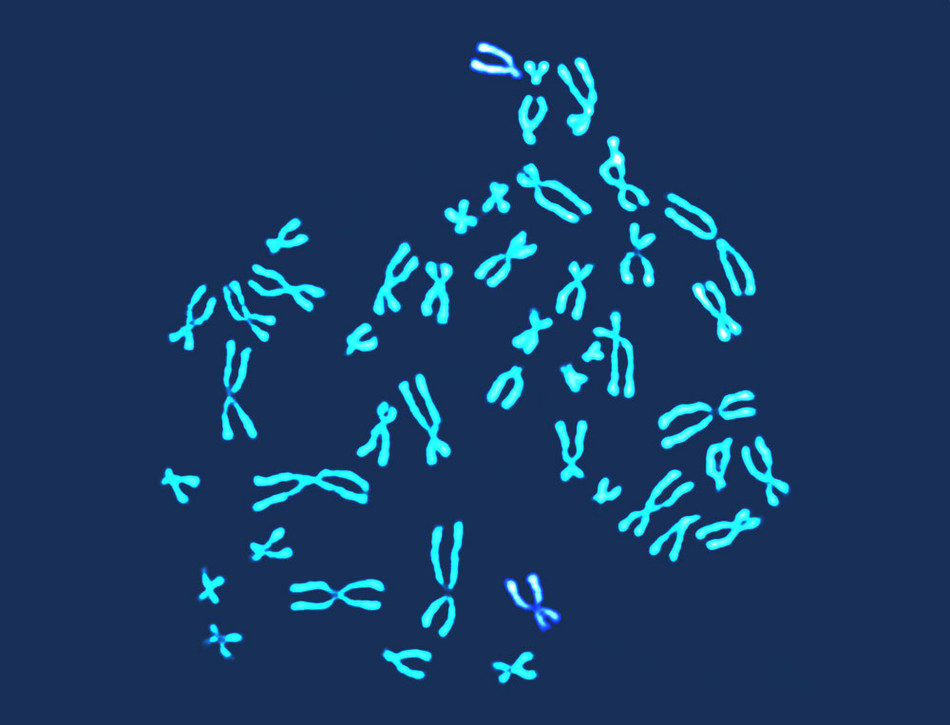
For decades, it has been commonplace for pregnant women to undergo a type of DNA testing that determines if a fetus is carrying any major genetic errors — like entire missing or extra chromosomes — that will cause severe birth defects. Called karyotyping, such analysis, which is also used to ensure the viability of embryos implanted in IVF procedures and to investigate the causes of miscarriages, has helped millions of women make more informed fertility choices. But results can take a week or longer to come back, and the testing costs upwards of $2,000, limiting its usefulness and making it unaffordable to many people.
Now a team of physicians led by Zev Williams, the director of the Columbia University Fertility Center, has developed a much faster and cheaper way to evaluate the genetic health of embryos and fetuses. The new technology, called STORK (for Short-read Transpore Rapid Karyotyping), can detect chromosomal abnormalities within two hours at a cost of less than $200.
“For a woman who’s wondering if her baby is healthy, every day she waits for an answer is agonizing, so the speed with which we’re able to generate results is extremely valuable,” says Williams. And because karyotyping isn’t always covered by insurance, he says, STORK’s affordability could make chromosomal analysis available to more women.
The culmination of nearly a decade of Columbia research, STORK consists of a compact handheld device that health-care workers can use to analyze DNA drawn from an embryo, amniotic fluid, placenta, or miscarried fetus. The tool’s key advantage is its automation: whereas karyotyping typically requires cells or tissues to be mailed out to specialized laboratories, where scientists inspect them under powerful microscopes, STORK enables chromosomal analysis to be performed at the point of care, such as in fertility centers or hospitals. The new system incorporates cutting-edge genomic-sequencing technology of a type that is typically used to spot mutations in individual genes but which the Columbia researchers have adapted to identify fetuses or embryos whose cells contain the wrong numbers of chromosomes — long threads of DNA that each contain hundreds or thousands of genes — or deformed or oddly sized DNA strands. “Because we’re not attempting to analyze the genome for small mutations but rather to detect catastrophic DNA-copying errors, our sequencing tool can operate at exceptional speed,” Williams says.
Like traditional karyotyping techniques, STORK can spot chromosomal errors that cause Down syndrome, fragile-X syndrome, and a number of even more severe congenital disorders, like Patau syndrome, which disrupts the development of major organs and typically proves fatal within a child’s first week. (Identifying a baby’s risk for medical conditions linked to mutations in single genes, like cystic fibrosis or Tay-Sachs disease, requires additional testing.)
In a recent letter published in the New England Journal of Medicine, Williams and his colleagues reported that STORK performed as reliably as standard karyotyping methods in analyzing 218 cell and tissue samples. The researchers have submitted their findings to the New York State Department of Health and expect STORK to be approved for widespread clinical use soon.
According to Williams, his team’s technology could prove especially beneficial to women who live in states that have banned abortion after the first three to five months of pregnancy, including Florida, North Carolina, and Utah. Delivering test results quickly to such women is critical, he says, because fetal cells cannot safely be biopsied for chromosomal analysis before the second trimester, which means that many women in these states are now learning that their babies are destined to have serious birth defects right around the time their access to abortion ends. “If a woman is considering ending a pregnancy in order to prevent terrible human suffering, she ought to be able to have that information as quickly as possible, for her health and safety,” Williams says.
For those pursuing in vitro fertilization, Williams says, STORK promises both convenience and savings. The current weeklong process of screening embryos for chromosomal abnormalities pushes back implantation a month, since it must be timed to the ovulation cycle. In the meantime, the woman must pay up to $7,000 to freeze the embryos being tested and undergo a separate embryo transfer. “With our method, an embryo can be analyzed in the morning and transferred in the afternoon,” he says.
And for women who miscarry, Williams says, STORK can help reveal why it happened. Approximately half of all miscarriages are caused by chromosomal abnormalities, he says, yet medical insurers typically will not pay for the karyotyping of miscarriage tissues unless a woman has previously lost one or more babies. As a consequence, many women are left wondering what went wrong. This is a significant problem, Williams says, because determining the cause of a miscarriage is the first step in preventing the next one.
“If you discover that a miscarried fetus had chromosomal issues, that’s actually reassuring news, in a sense, because such errors tend to occur randomly. This means that a woman who conceives again is just as likely to have a healthy baby as she would have been if that prior loss had not occurred,” he says. “That’s important information to be able to deliver to people, because our research has shown that when miscarriages are not explained, women tend to blame themselves. They’ll say, ‘Oh, it was because of that deadline at work, a stressful argument that I had, or lifting something heavy.’ On the other hand, if you learn that a miscarried fetus was genetically healthy, now you know as a physician to look for other explanations. And there are a number of very treatable medical conditions that can interfere with a woman’s ability to carry a baby to term. You just have to know to look for them in order to solve them.”